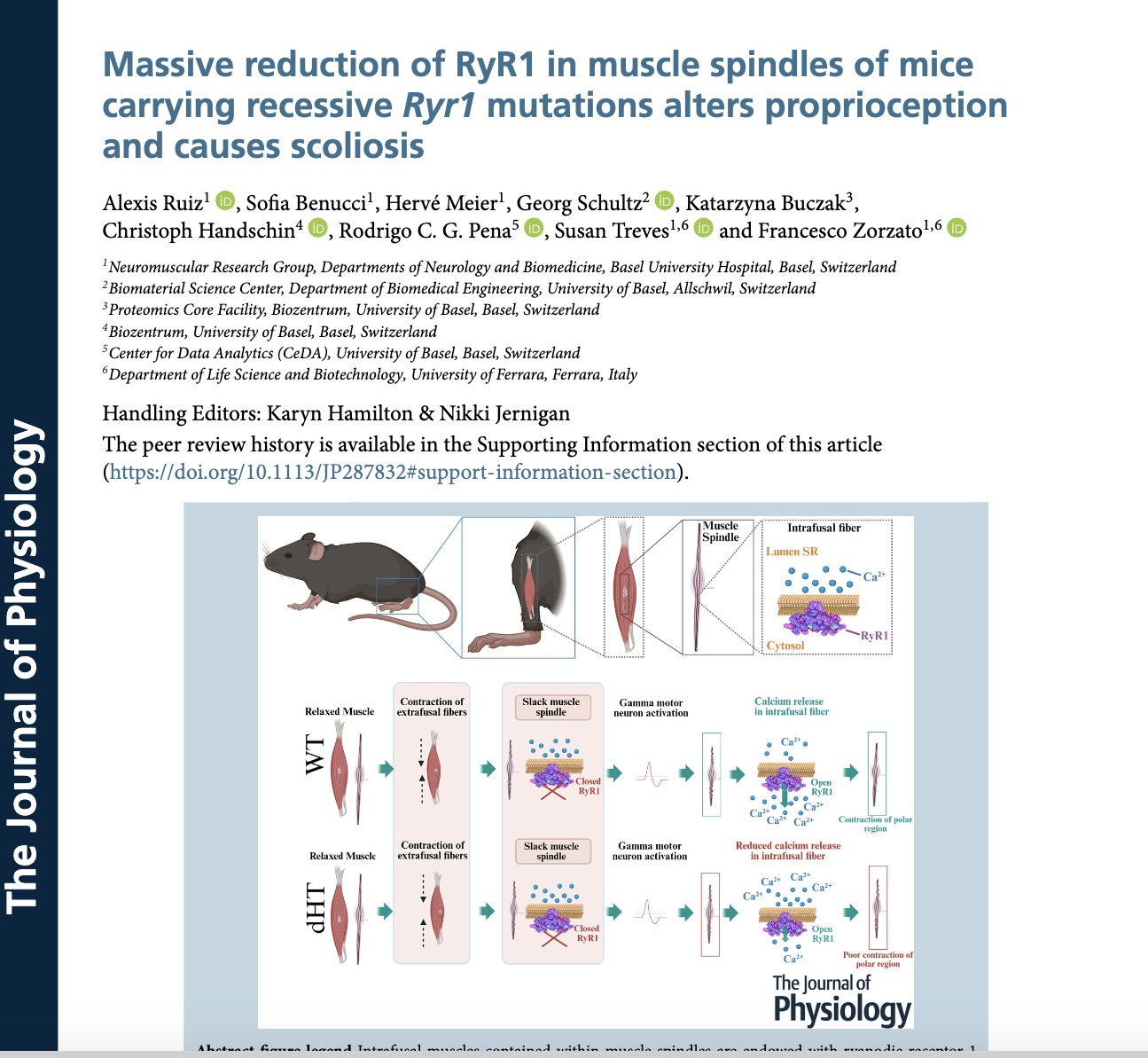
Abstract
Muscle spindles are stretch receptors lying deep within the muscle belly involved in detecting changes in muscle length and playing a fundamental role in motor control, posture and synchronized gait. They are made up of an external capsule surrounding three to five intrafusal muscle fibres and a nuclear bag complex. Dysfunction of muscle spindles leads to abnormal proprioceptor function, which has been linked to aberrant bone and cartilage development, scoliosis, kyphosis and joint contractures. RYR1, the gene encoding the calcium release channel of the sarcoplasmic reticulum, is the most common target of mutations linked to human congenital myopathies, a condition often accompanied by skeleton alterations and joint contractures. So far, the link between RYR1 mutations, altered muscle spindles and skeletal defects has not been investigated. To this end, we investigated heterozygous mice carrying recessive Ryr1 mutations isogenic to those present in a severely affected child. Here, we show that: (i) the RyR1 protein localizes to the polar regions of intrafusal fibres and exhibits a doubled row distribution pattern, typical for junctional sarcoplasmic reticulum proteins; (ii) muscle spindles of compound heterozygous mice show structural defects; and (iii) in intrafusal muscle fibres from dHT mice, RyR1-mediated Ca2+ release is significantly impaired and RyR1 protein content is reduced by 54%. These results support the hypothesis that Ryr1 mutations not only affect the function of extrafusal muscles, but also might affect that of intrafusal muscles. The latter may be one of the underlying causes of skeletal abnormalities seen in patients affected by recessive RYR1 mutations.